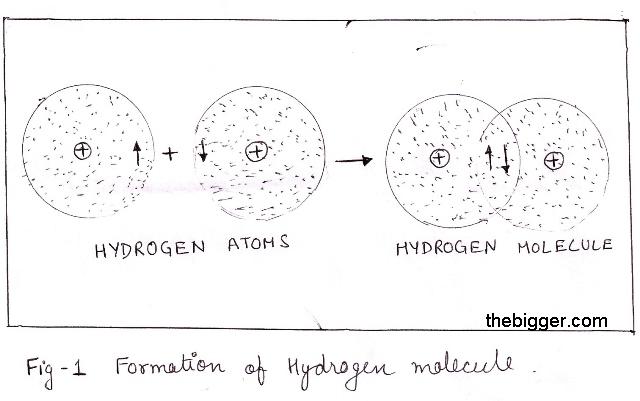
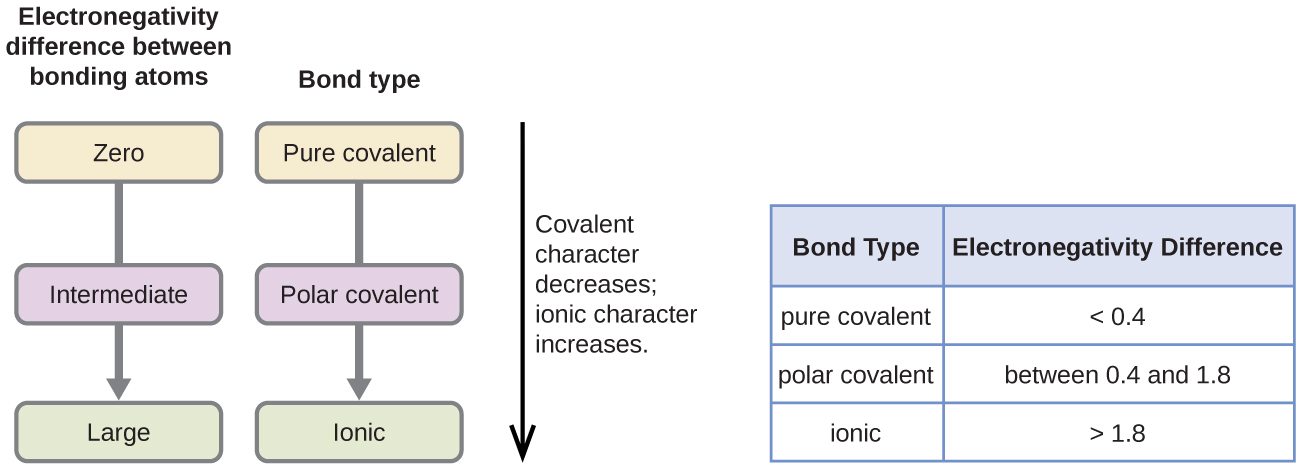
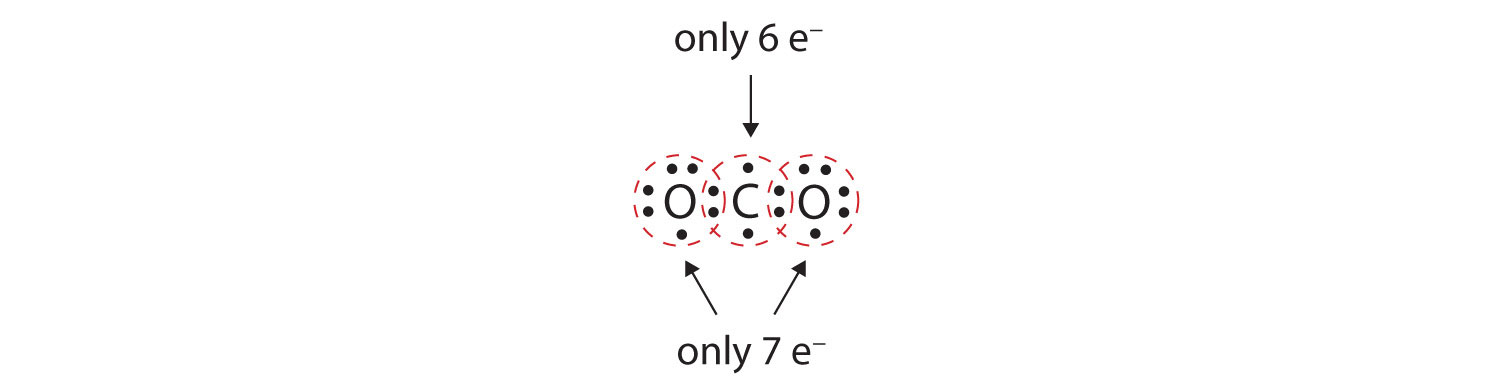
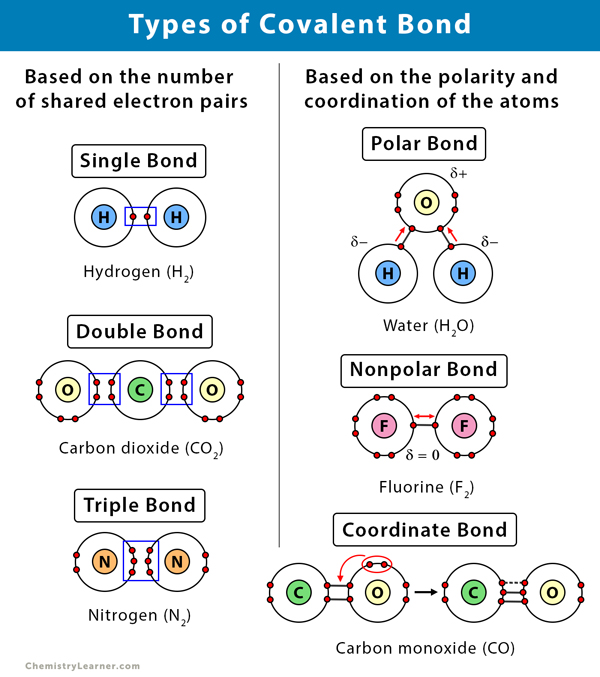
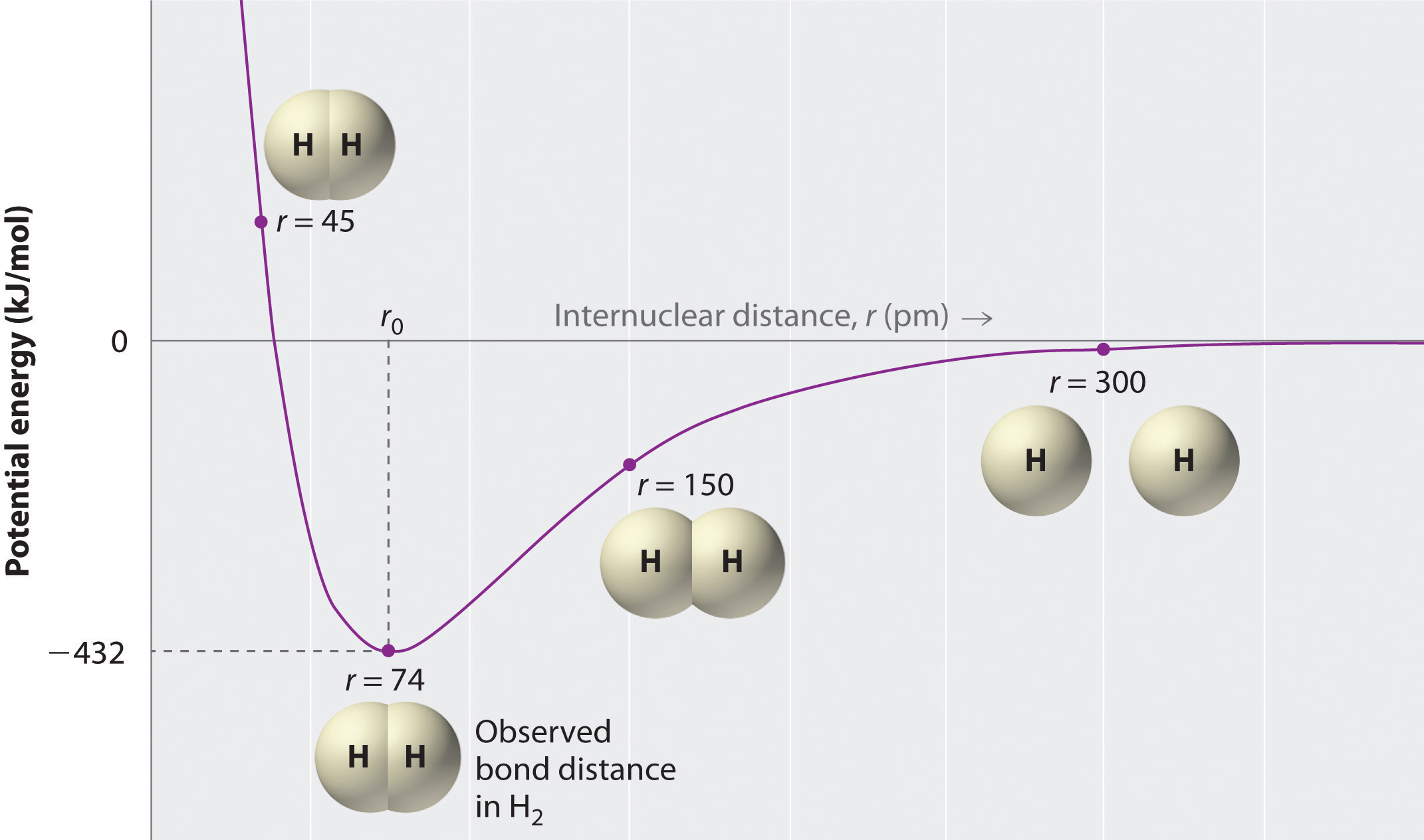
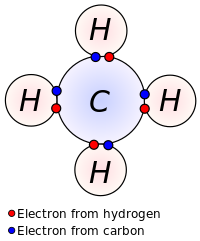
SOLVED: Tbe strength of a covalent bond depends upon the size of the atoms and the bond order In general short bonds are strong bonds For each pair of covalently bonded atoms,

SOLVED: Question 8 A(n) is composed of long chains of covalently bonded atoms polymer polar covalent molecule London dispersion force ionic compound Flag this Question Question 9 In general, if a gas